

Modern vaccine development takes advantage of technological advances and scientific understanding of the immune system and host-pathogen interactions. At Intravacc, the creation of a vaccine begins with viral or bacterial strain design.
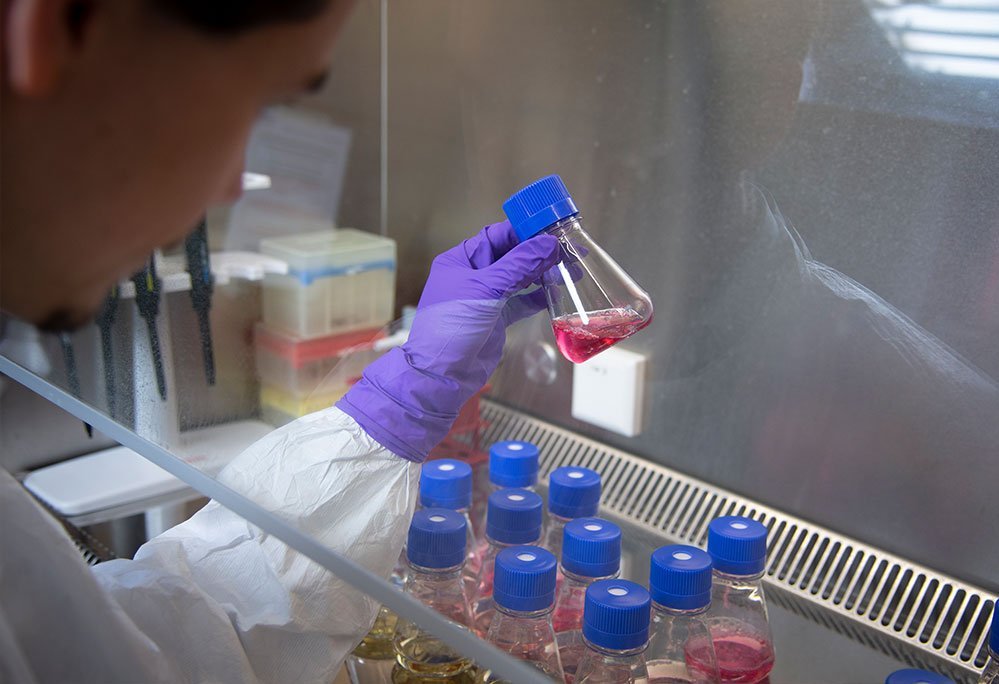
For our viral vaccine candidates, we aim to rescue virus strains from plasmid DNA. To achieve this, we use optimized production cell lines and virus strains in the development of inactivated viruses, live-attenuated viruses, virus-like particles, and alternatives like single-round infectious particles, replicons, and virus vectors.
Our flexible technologies for the development of bacterial vaccines includes inactivated bacteria, bacterial toxins, conjugate vaccines, and our proprietary Outer Membrane Vesicles (OMV) platform.

We take advantage of our existing vaccine platforms Cell-Vacc, OMV-Vacc, and Con-Vacc to develop therapeutic vaccines for our clients. From conjugate vaccines for treatment of neurodegenerative diseases like a seldom type of amyotrophic lateral sclerosis, to oncolytic viruses to treat different types of cancer.
We select vaccine antigens via bioinformatics or forced evolution, in silico design, and subsequent construction. In this process, we optimize safety, immunogenicity (such as level of expression and epitope design) and intrinsic antigen stability.


To ensure GMP readiness, we genetically modify our bacterial strains without selection markers in the final production strain. Our viral strains are carefully vetted for purity and quality criteria. Finally, we emphasize production processes that are free of animal components.
You can send us an email:
info@intravacc.nl
Reach out to Business Development:
BD@intravacc.nl
Or pick up the phone:
+31 30 792 03 00
You can also just fill out the contact form on the right.
We look forward to hearing from you!
